Signal Transduction Pathways
Sakshi Education
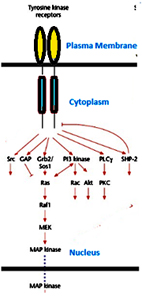
Binding of a signaling molecule to the extracellular domains of the receptor leads to its activation. Activation of plasma membrane receptors occurs mainly by induced conformational changes or dimerization or oligomerization which subsequently initiates a sequence of events leading to a signaling pathway. This requires the role of various signaling molecules which contain different types of conserved domains arranged in a modular fashion. These modular domains define the kind of target molecules they can interact with and thus impart a degree of specificity to the signaling mechanisms. They act as docking sites for both the upstream and downstream components in the pathway.
Receptor Tyrosine Kinases mediated signaling pathways
Cell surface receptors play a dual role of not only act as receptors but act as enzymes. Binding of ligands to the receptors activates the inherent enzymatic activity. Receptor Tyrosine kinases make the largest of such as class. Signaling molecules with domains like SH2 (src homology 2) and PTB (phosphotyrosine binding) domains are involved in these pathways. They recognize phosphorylated tyrosine residues in defined environments. SH2 domains specifically recognize motifs based on a phosphorylated tyrosine on the C- terminal sequences, whereas PTB domains recognize motifs based on a phosphorylated tyrosine on the N- terminal sequences. Each one of the SH2 or PTB domain-containing signaling molecules binds-to auto phosphorylated receptors or receptor-associated molecules potentially initiate a signaling pathway. Multiple SH2 domain proteins can bind simultaneously to an activated receptor.
Phospholipids in the membrane are involved in two of such pathways. Phosphatidyl inositol 3’-kinase (PI3K) phosphorylates phosphatidyl inositol 4,5-bisphosphate (PIP2 or PtdIns(4,5)P2) to PtdIns(3,4,5)P3, which leads to activation of the Serine/ threonine kinase Akt and the small GTPase Rac,mediating anti-apoptosis and reorganization of actin filaments and cell migration,respectively. PI3K consists of both a catalytic subunit and an SH2domain-containing regulatory subunit, upon binding of the regulatory subunit to phosphorylated tyrosine residues, e.g. inactivated receptors, the kinase activity of the catalytic subunit is activated. Insulin like growth factors use this pathway.
In an another pathway the membrane phospholipid involved is Phosphatidyl inositol bis phosphate (PIP2) which is cleaved by phospholipase C-?(PLC?) to diacylglycerol (DAG) and inositol 1,4,5 tris phosphate (IP3). IP3 activates members of the protein kinaseC family and mobilizes Ca2+from intracellular stores, respectively. PLC? has intrinsic SH2 domains with which it binds to activated receptors. It is then phosphorylated on a specific tyrosine residue, which activates the catalytic activity.
MAP Kinase Pathways
Mitogen activated protein (MAP) kinase cascade is one of the common and important signaling pathway triggered by RTKs. Cascade of reactions take place due to the presence of three Serine/ threonine kinases.The pathway is initiated by the activation of the small GTPase, Ras and the Erk MAP kinase cascade, leading to cell proliferation. Ras is activated by the nucleotide exchange molecule Sos1, which forms a complex with the adaptor molecule Grb2. Signaling molecules like Grb2 lack an intrinsic kinase activity but contain domains for binding of different molecules and thus act as adaptor proteins. They play a role of coordinating different molecules involved in the signaling mechanism. The interaction of the Grb2/Sos1complex with Ras on the inner surface of the plasma membrane leads to activation of Ras which subsequently binds directly to the the first of the three Serine/threonine kinases, Raf. Raf phosphorylates and activates the second Serine/threonine kinase, MEK serine/ threonine kinase which in turn, phosphorylates and activates the MAP kinase Erk (extracellular receptor regulated kinase),which then is translocated into the nucleus,where it regulates the activity of specific transcription factors through phosphorylation.
Members of the Src family of tyrosine kinases appear to be important for the mitogenic response of certain growth stimuli. Src contains three domains a kinase domain, a SH2 domain and a SH3 domain. In its inactive form, Src is phosphorylated on a C-terminal tyrosine residue, which is involved in an intramolecular interaction with its SH2 domain. Src is activated by the binding of the SH2 domain of Src to a phosphorylated tyrosine residue of an activated tyrosine kinase receptor,together with dephosphorylation of the C-terminal tyrosine and phosphorylation of other tyrosine residues.
JAK-STAT Pathways
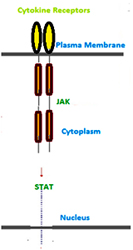 Cytokine receptors mediate downstream signaling pathways constituted by STAT (signal transducer and activator of transcription) molecules which act as both the signal transducer and transcription factor. These pathways by employing a strategy to directly activate the transcription factor minimize the role of second messengers and also hasten the response process. Cytokine receptors lack intrinsic kinase activity and hence employ tyrosine kinases of the JAK family for phosphorylation. STAT molecules bind to phosphorylated tyrosine residues via their SH2 domain. The SH2domain in STATs instead mediates an intermolecular binding within a STAT dimer. Dimerized STAT molecules are translocated to the nucleus where they regulate the transcription of specific genes. Thus, SH2 domain-mediated interactions with activated receptors lead to activation of different signaling pathways with different mechanisms. The docking of a signaling molecule can lead to its phosphorylation and activation, such as in the cases of PLC?, Src and STATs.
Cytokine receptors mediate downstream signaling pathways constituted by STAT (signal transducer and activator of transcription) molecules which act as both the signal transducer and transcription factor. These pathways by employing a strategy to directly activate the transcription factor minimize the role of second messengers and also hasten the response process. Cytokine receptors lack intrinsic kinase activity and hence employ tyrosine kinases of the JAK family for phosphorylation. STAT molecules bind to phosphorylated tyrosine residues via their SH2 domain. The SH2domain in STATs instead mediates an intermolecular binding within a STAT dimer. Dimerized STAT molecules are translocated to the nucleus where they regulate the transcription of specific genes. Thus, SH2 domain-mediated interactions with activated receptors lead to activation of different signaling pathways with different mechanisms. The docking of a signaling molecule can lead to its phosphorylation and activation, such as in the cases of PLC?, Src and STATs.
Serine Threonine Kinase Receptor Mediated Signaling Pathways
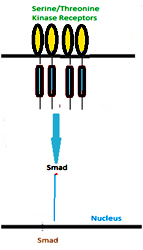 Serine/threonine kinase receptors also cause direct activation of transcription factors similar to JAK-STAT pathways. These activated transcription factors translocate to the nucleus to activate the transcription of target genes. Upon binding of a ligand the receptor gets activated and further activates the effector molecules by causing phosphorylation. Thus receptor activated Smad (R-Smad) molecules are phosphorylated in-SXS motifs in their C-termini. There is a selectivity in the recognition of specific Smad molecules by certain type I receptors; in particular, complementarity between a loop in the kinase domain of the receptor (the L45 loop) and a specific structure in the carboxy-terminal domain of Smads (the L3 loop) is important. Phosphorylation of R-Smads triggers interaction with common mediator Smad4; the Smad complexes are then translocated to the nucleus where they, in cooperation with other transcription factors, regulate specific genes.
Serine/threonine kinase receptors also cause direct activation of transcription factors similar to JAK-STAT pathways. These activated transcription factors translocate to the nucleus to activate the transcription of target genes. Upon binding of a ligand the receptor gets activated and further activates the effector molecules by causing phosphorylation. Thus receptor activated Smad (R-Smad) molecules are phosphorylated in-SXS motifs in their C-termini. There is a selectivity in the recognition of specific Smad molecules by certain type I receptors; in particular, complementarity between a loop in the kinase domain of the receptor (the L45 loop) and a specific structure in the carboxy-terminal domain of Smads (the L3 loop) is important. Phosphorylation of R-Smads triggers interaction with common mediator Smad4; the Smad complexes are then translocated to the nucleus where they, in cooperation with other transcription factors, regulate specific genes.
Regulation of Signaling Mechanisms
Receptor Tyrosine Kinases mediated signaling pathways
Cell surface receptors play a dual role of not only act as receptors but act as enzymes. Binding of ligands to the receptors activates the inherent enzymatic activity. Receptor Tyrosine kinases make the largest of such as class. Signaling molecules with domains like SH2 (src homology 2) and PTB (phosphotyrosine binding) domains are involved in these pathways. They recognize phosphorylated tyrosine residues in defined environments. SH2 domains specifically recognize motifs based on a phosphorylated tyrosine on the C- terminal sequences, whereas PTB domains recognize motifs based on a phosphorylated tyrosine on the N- terminal sequences. Each one of the SH2 or PTB domain-containing signaling molecules binds-to auto phosphorylated receptors or receptor-associated molecules potentially initiate a signaling pathway. Multiple SH2 domain proteins can bind simultaneously to an activated receptor.
Phospholipids in the membrane are involved in two of such pathways. Phosphatidyl inositol 3’-kinase (PI3K) phosphorylates phosphatidyl inositol 4,5-bisphosphate (PIP2 or PtdIns(4,5)P2) to PtdIns(3,4,5)P3, which leads to activation of the Serine/ threonine kinase Akt and the small GTPase Rac,mediating anti-apoptosis and reorganization of actin filaments and cell migration,respectively. PI3K consists of both a catalytic subunit and an SH2domain-containing regulatory subunit, upon binding of the regulatory subunit to phosphorylated tyrosine residues, e.g. inactivated receptors, the kinase activity of the catalytic subunit is activated. Insulin like growth factors use this pathway.
In an another pathway the membrane phospholipid involved is Phosphatidyl inositol bis phosphate (PIP2) which is cleaved by phospholipase C-?(PLC?) to diacylglycerol (DAG) and inositol 1,4,5 tris phosphate (IP3). IP3 activates members of the protein kinaseC family and mobilizes Ca2+from intracellular stores, respectively. PLC? has intrinsic SH2 domains with which it binds to activated receptors. It is then phosphorylated on a specific tyrosine residue, which activates the catalytic activity.
MAP Kinase Pathways
Mitogen activated protein (MAP) kinase cascade is one of the common and important signaling pathway triggered by RTKs. Cascade of reactions take place due to the presence of three Serine/ threonine kinases.The pathway is initiated by the activation of the small GTPase, Ras and the Erk MAP kinase cascade, leading to cell proliferation. Ras is activated by the nucleotide exchange molecule Sos1, which forms a complex with the adaptor molecule Grb2. Signaling molecules like Grb2 lack an intrinsic kinase activity but contain domains for binding of different molecules and thus act as adaptor proteins. They play a role of coordinating different molecules involved in the signaling mechanism. The interaction of the Grb2/Sos1complex with Ras on the inner surface of the plasma membrane leads to activation of Ras which subsequently binds directly to the the first of the three Serine/threonine kinases, Raf. Raf phosphorylates and activates the second Serine/threonine kinase, MEK serine/ threonine kinase which in turn, phosphorylates and activates the MAP kinase Erk (extracellular receptor regulated kinase),which then is translocated into the nucleus,where it regulates the activity of specific transcription factors through phosphorylation.
Members of the Src family of tyrosine kinases appear to be important for the mitogenic response of certain growth stimuli. Src contains three domains a kinase domain, a SH2 domain and a SH3 domain. In its inactive form, Src is phosphorylated on a C-terminal tyrosine residue, which is involved in an intramolecular interaction with its SH2 domain. Src is activated by the binding of the SH2 domain of Src to a phosphorylated tyrosine residue of an activated tyrosine kinase receptor,together with dephosphorylation of the C-terminal tyrosine and phosphorylation of other tyrosine residues.
JAK-STAT Pathways
 Cytokine receptors mediate downstream signaling pathways constituted by STAT (signal transducer and activator of transcription) molecules which act as both the signal transducer and transcription factor. These pathways by employing a strategy to directly activate the transcription factor minimize the role of second messengers and also hasten the response process. Cytokine receptors lack intrinsic kinase activity and hence employ tyrosine kinases of the JAK family for phosphorylation. STAT molecules bind to phosphorylated tyrosine residues via their SH2 domain. The SH2domain in STATs instead mediates an intermolecular binding within a STAT dimer. Dimerized STAT molecules are translocated to the nucleus where they regulate the transcription of specific genes. Thus, SH2 domain-mediated interactions with activated receptors lead to activation of different signaling pathways with different mechanisms. The docking of a signaling molecule can lead to its phosphorylation and activation, such as in the cases of PLC?, Src and STATs.
Cytokine receptors mediate downstream signaling pathways constituted by STAT (signal transducer and activator of transcription) molecules which act as both the signal transducer and transcription factor. These pathways by employing a strategy to directly activate the transcription factor minimize the role of second messengers and also hasten the response process. Cytokine receptors lack intrinsic kinase activity and hence employ tyrosine kinases of the JAK family for phosphorylation. STAT molecules bind to phosphorylated tyrosine residues via their SH2 domain. The SH2domain in STATs instead mediates an intermolecular binding within a STAT dimer. Dimerized STAT molecules are translocated to the nucleus where they regulate the transcription of specific genes. Thus, SH2 domain-mediated interactions with activated receptors lead to activation of different signaling pathways with different mechanisms. The docking of a signaling molecule can lead to its phosphorylation and activation, such as in the cases of PLC?, Src and STATs.Serine Threonine Kinase Receptor Mediated Signaling Pathways
 Serine/threonine kinase receptors also cause direct activation of transcription factors similar to JAK-STAT pathways. These activated transcription factors translocate to the nucleus to activate the transcription of target genes. Upon binding of a ligand the receptor gets activated and further activates the effector molecules by causing phosphorylation. Thus receptor activated Smad (R-Smad) molecules are phosphorylated in-SXS motifs in their C-termini. There is a selectivity in the recognition of specific Smad molecules by certain type I receptors; in particular, complementarity between a loop in the kinase domain of the receptor (the L45 loop) and a specific structure in the carboxy-terminal domain of Smads (the L3 loop) is important. Phosphorylation of R-Smads triggers interaction with common mediator Smad4; the Smad complexes are then translocated to the nucleus where they, in cooperation with other transcription factors, regulate specific genes.
Serine/threonine kinase receptors also cause direct activation of transcription factors similar to JAK-STAT pathways. These activated transcription factors translocate to the nucleus to activate the transcription of target genes. Upon binding of a ligand the receptor gets activated and further activates the effector molecules by causing phosphorylation. Thus receptor activated Smad (R-Smad) molecules are phosphorylated in-SXS motifs in their C-termini. There is a selectivity in the recognition of specific Smad molecules by certain type I receptors; in particular, complementarity between a loop in the kinase domain of the receptor (the L45 loop) and a specific structure in the carboxy-terminal domain of Smads (the L3 loop) is important. Phosphorylation of R-Smads triggers interaction with common mediator Smad4; the Smad complexes are then translocated to the nucleus where they, in cooperation with other transcription factors, regulate specific genes.Regulation of Signaling Mechanisms
Julian Downward (2008) Nature Medicine 14, 1315 – 1316.
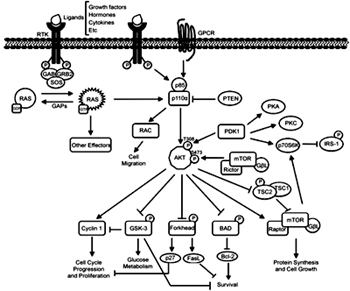
Esther Castellano and Julian Downward (2011) Genes Cancer; 2(3): 261–274.
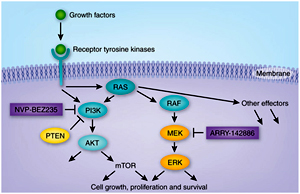
Although signal transduction appears to occur simply through activation of a number of parallel pathways, they exhibit an extensive crosstalk between the components of the different pathways. For example, PI3K and Ras, major signaling pathways downstream of tyrosine kinase receptors, interact physically and activate each other.
Moreover, STAT-molecules, which are typically activated by cytokine receptors, are also activated by certain tyrosine kinase receptors. In addition the typical pathways induced downstream of tyrosine kinase receptors, e.g. PLC?, PI3K and Src, are also activated by cytokine receptors, or by Integrins. Various studies have also shown that G protein-coupled receptors and pathways downstream of tyrosine kinase receptors are also interlinked.
Thus different pathways positively regulate each other by means of activation. Nevertheless there are pathways that regulate each other negatively. Inactivating crosstalk occur when MAP kinase, which is activated downstream of tyrosine kinase or cytokine receptors inhibits Smad by phosphorylation. Thus intracellular signaling pathways activate a network of pathways rather than a linear pathway. Cell exhibits different strategies to regulate signaling mechanisms. Most important feature of regulation is to control the magnitude of signaling molecules generated. This occurs mainly by different inhibitory mechanisms mentioned below:
Although signal transduction appears to occur simply through activation of a number of parallel pathways, they exhibit an extensive crosstalk between the components of the different pathways. For example, PI3K and Ras, major signaling pathways downstream of tyrosine kinase receptors, interact physically and activate each other.
Moreover, STAT-molecules, which are typically activated by cytokine receptors, are also activated by certain tyrosine kinase receptors. In addition the typical pathways induced downstream of tyrosine kinase receptors, e.g. PLC?, PI3K and Src, are also activated by cytokine receptors, or by Integrins. Various studies have also shown that G protein-coupled receptors and pathways downstream of tyrosine kinase receptors are also interlinked.
Thus different pathways positively regulate each other by means of activation. Nevertheless there are pathways that regulate each other negatively. Inactivating crosstalk occur when MAP kinase, which is activated downstream of tyrosine kinase or cytokine receptors inhibits Smad by phosphorylation. Thus intracellular signaling pathways activate a network of pathways rather than a linear pathway. Cell exhibits different strategies to regulate signaling mechanisms. Most important feature of regulation is to control the magnitude of signaling molecules generated. This occurs mainly by different inhibitory mechanisms mentioned below:
- Inhibition of signaling molecules from binding to the receptors
Certain secreted proteins bind to TGFß family members and thereby inhibit from binding to receptors preventing the activation of receptors.
- Employing decoy receptors
Certain receptors act as imitating models of the true receptors and allow binding of the signaling molecules or ligands but does not generate any downstream signal. Such receptors are termed as Decoy receptors. The role of such receptors is mostly implicated in controlling the strength of the signal by reducing the number of ligands actually binding to true receptors. They do not completely inhibit activation of true receptors.
- Blocking the receptors
Chaperone like proteins are reported to be involved in binding to TGFß and FK-BP 12 receptors and block them. Thus they physically exclude the signaling molecules from binding to the receptors.
- By inhibitory phosphorylation of signaling molecules
R-SMADs belonging to Serine/threonine Kinase receptor pathways involved in cell growth inhibition, differentiation and apoptosis are inactivated due to inhibitory phosphorylation by MAP Kinase belonging to Tyrosine Kinase receptor pathway.
- By prevention of transcription of the target genes
Activated receptors phosphorylate effector molecules which translocate into the nucleus and initiate transcription of the target genes. Certain inhibitory proteins bind to activators and co-activators required for transcription initiation by the RNA polymerase and thereby prevent gene expression by the effector molecules.
- Induction of inhibitory molecules
Inhibitory molecules like SMAD 6 and SMAD 7 compete with the signal molecules belonging to the same family -R SMADs for binding to the receptors and thereby prevent phosphorylation and activation of the R SMADs.
- Induction of inhibitory signals
In certain cases ligand binding to specific receptors can not only induce stimulatory signals for activation of the pathway but also induce inhibitory signals simultaneously. For instance, activation of Ras by the PDGF receptor occurs by binding of the Grb2/Sos1 complex directly or indirectly to the receptor. However, the PDGFb receptor also binds a GTPase-activating protein (GAP) for Ras, which performs the opposite reaction by catalyzing the conversion of the active GTP Ras to the inactive GDP Ras.
- Proteolytic degradation of the signaling molecules.
Effector molecules involved in tyrosine kinase receptors and Serine/ threonine kinase receptors are recognized by ubiquitin ligases and thereby tagged for degradation in proteasomes. Such proteolytic degradation of the molecules serves dual purposes. Primarily for controlling the basal level of signaling molecules and secondly as a shut-off mechanism wherein activated signaling molecules are deactivated.
- Esther Castellano and Julian Downward (2011) More than just Another Effector Pathway. Genes Cancer; 2(3): 261–274.
- Julian Downward (2008) Targeting RAS and PI3K in lung cancer. Nature Medicine: 14, 1315 – 1316.
- Ubersax, J. A., & Ferrell, Jr., J. E. (2007) Mechanisms of Specificity in Protein Phosphorylation. Nature Reviews Molecular Cell Biology 8, 530–541
- Hunter T (2000) Signaling-2000 and Beyond. Cell: 113-127.
- Pawson T and Scott JD (1997) Signaling Through Scaffold, Anchoring and Adaptor Proteins. Science 278:2075-2080.
Published date : 14 May 2014 02:23PM





