Intermediate: 1st Year Chemistry Model Paper -2
Sakshi Education
IPE MODEL PAPER-II
CHEMISTRY
Time- 3 Hrs Max. Marks: 60
Section – A
10 x 2 = 20M
I. Very short answer type questions.
i) Answer any 10 Questions. ii) Each question carries two marks.
1. Define Osmotic pressure.
2. Write the structure of XeO3.
3. How do you distinguish between crystal lattice and unit cell.
4. What is rate law? Illustrate with an example?
5. What is Brownian movement.
6. Electron gain. Enthalpy of F2 is less than Cl2- explain.
7. What is mischmetal? Give its composition and uses?
8. Write the ring structure and open chain structure of glucose?
9. Write the structures of the following compounds.
a) 2-Chloro-3-methyl pentane b)2-Bromo-3-methyl Hexane
10. Give structures of A,B and C in the following reaction
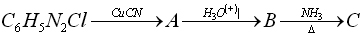
i) Answer any 10 Questions. ii) Each question carries two marks.
1. Define Osmotic pressure.
2. Write the structure of XeO3.
3. How do you distinguish between crystal lattice and unit cell.
4. What is rate law? Illustrate with an example?
5. What is Brownian movement.
6. Electron gain. Enthalpy of F2 is less than Cl2- explain.
7. What is mischmetal? Give its composition and uses?
8. Write the ring structure and open chain structure of glucose?
9. Write the structures of the following compounds.
a) 2-Chloro-3-methyl pentane b)2-Bromo-3-methyl Hexane
10. Give structures of A,B and C in the following reaction
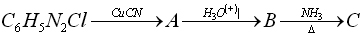
Section – B
6 x 4 = 24M
II. Short answer type questions.
i) Attempt any six Questions. ii) Each question carries four marks.
11. Write the IUPAC names of the following coordination compounds?
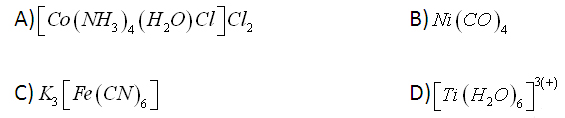
12. Define mole fraction. The vapour pressure of pure Benzene at a certain temperature is 0.850 bar. A non volatile, non-Electrolyte solid weighing 0.5g when added to 39g C6H6. Vapour pressure of the solution then 0.845 bar. What is the molar mass of the solid substance.
13. What are physisorption and chemsorption? Write any four differences between physisorption and chemisorption?
14. Explain a)Kolbe’s reaction b)Acidity of phenol
15. How is ammonia manufactured by Haber’s process?
16. a)Define Frenkel defect.
b)An X-ray from beam (wave length=70.93pm) was scattered by a crystalline solid. The angle (2q) of diffraction for a second order reflection is 14’16’. Calculate the distance between parallel planes of atoms from which the scattered beam appears to have been reflected.
17.
i) Attempt any six Questions. ii) Each question carries four marks.
11. Write the IUPAC names of the following coordination compounds?
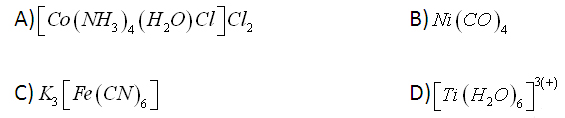
12. Define mole fraction. The vapour pressure of pure Benzene at a certain temperature is 0.850 bar. A non volatile, non-Electrolyte solid weighing 0.5g when added to 39g C6H6. Vapour pressure of the solution then 0.845 bar. What is the molar mass of the solid substance.
13. What are physisorption and chemsorption? Write any four differences between physisorption and chemisorption?
14. Explain a)Kolbe’s reaction b)Acidity of phenol
15. How is ammonia manufactured by Haber’s process?
16. a)Define Frenkel defect.
b)An X-ray from beam (wave length=70.93pm) was scattered by a crystalline solid. The angle (2q) of diffraction for a second order reflection is 14’16’. Calculate the distance between parallel planes of atoms from which the scattered beam appears to have been reflected.
17.
a) What is the effect of temperature on the rate constant?
b) Calculate the half-life of first order reaction whose rate constant is 200S-1.
b) Calculate the half-life of first order reaction whose rate constant is 200S-1.
18. Defined the following as related to proteins.
a) Peptide linkage b) Primary structure c) Denaturation
a) Peptide linkage b) Primary structure c) Denaturation
Section – C
2 x 8 = 16M
III. Long answer type questions.
i) Attempt any two Questions. ii) Each question carries eight marks.
19. How is chlorine prepared in the laboratory? How does it react with the following?
a)Iron b)Acidified FeSO4 c) Na2S2O3 d) H2s
20. a)Define order of reaction? Illustrate your answer with an example.
b)Define molecularity of a reaction? Illustrate with an example.
c)What is half-life period? Write equation useful to calculate half-lifes for zero and first order reaction.
21. Explain a)crossed aldol condensation b)Reimer-Tiemann’s reaction
c)Sandmayer’s reaction d)H.V.Z. reaction
*****
Published date : 11 Jul 2021 05:21PM













